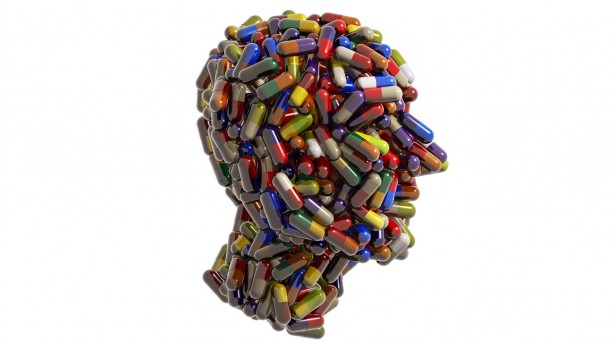
“Drugs companies publish only a fraction of their results and keep much of the information to themselves, but regulators want to ban the practice. If companies published all of their clinical trials data, independent scientists could reanalyse their results and check companies’ claims about the safety and efficacy of drugs. Under proposals being thrashed out in Europe, drugs companies would be compelled to release all of their data, including results that show drugs do not work or cause dangerous side-effects. The latest strategy shows how patient groups – many of which receive some or all of their funding from drugs companies – have been brought into the battle.”
Related posts:
China's military makes move into Africa
CNN: "We've Received Confirmation He Was Unarmed When The FBI Shot Him 7 Times Once In The Head"
What My 6-Year-Old Son and I Endured in Family Detention
Meet the Bitcoin Millionaires
Canadian family's $500K inheritance seized by U.S. border officials
Electric Carmaker Struggles as Its Production Lags
European envoy meets with Egyptian ex-president Morsi
New York auction houses celebrate after billion-dollar sales week
Russians discover four vats of embryos during stroll in the woods
North and South Korea prepare to negotiate directly after years of mistrust
U.S. ‘very disappointed’ by Russian ban on U.S. meat
West Point sergeant accused of filming naked female cadets without their consent
South Korean Activists Send Leaflets to North Korea
Too cute to kill? Americans split on whether or not to kill suburban deer
Bitcoin Currency Use Impeded by IRS Property Treatment
